














| Still deciding? Get samples of $ ! US$ 10/g Request Sample |

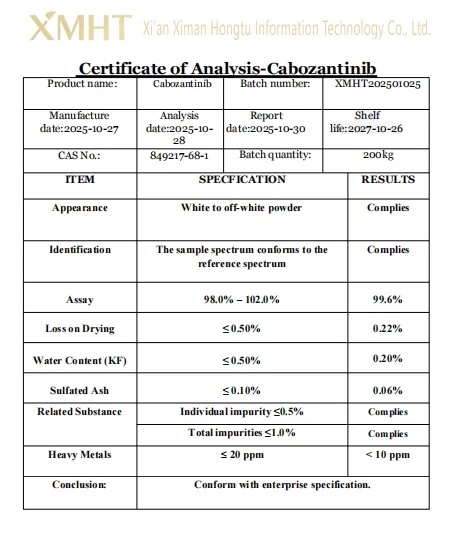
| Product Name | Cabozantinib |
| CAS No. | 849217-68-1 |
| Certification | GMP, HSE, ISO 9001, USP, BP |
| Purity | >99% |
| Appearance | White Powder |
| Molecular Formula | C28H24FN3O5 |
| Molecular Weight | 501.514 |
| Density | 1.397 |
| Boiling Point | 758.075 °C |
| Grade | Pharmaceutical Grade |
| Shelf Life | 2 Years |
| Test Method | HPLC |
| Production Capacity | 200kg/Month |


Cabozantinib is an oral multi-targeted tyrosine kinase inhibitor that simultaneously inhibits multiple kinases closely involved in tumor growth, angiogenesis, and metastasis, such as MET, VEGFR2, RET, and AXL. By blocking these key signaling pathways, it inhibits tumor cell proliferation, induces apoptosis, and reduces tumor angiogenesis.


Cabozantinib is indicated for the treatment of advanced renal cell carcinoma, hepatocellular carcinoma, and radioactive iodine-refractory differentiated thyroid cancer. This broad mechanism of action allows it to exert antitumor effects through several coordinated pathways: it inhibits oncogenic signaling that drives tumor growth and survival, blocks tumor angiogenesis, and modulates the tumor microenvironment.








 SJ medical
SJ medical