














| Still deciding? Get samples of $ ! US$ 50/kg Request Sample |


Lapatinib is an oral small-molecule tyrosine kinase inhibitor. It targets specific receptors and is primarily used in the treatment of advanced breast cancer, particularly when disease has progressed despite prior therapies. It works by blocking intracellular signaling pathways that promote tumor cell proliferation and survival.
| Product Name | Lapatinib |
| Appearance | White to Off-White Crystalline Powder |
| CAS No. | 231277-92-2 |
| Molecular Formula | C29H26ClFN4O4S |
| Molecular Weight | 581.057543 |
Lapatinib acts as a dual-targeted tyrosine kinase inhibitor. It inhibits the tyrosine kinase activity of both HER1 (EGFR) and HER2 receptors. By binding to the intracellular kinase domains, it blocks signaling pathways like PI3K-AKT and RAS-MAPK, effectively reducing tumor cell proliferation and inducing apoptosis.
Key benefits include its ability to cross the blood-brain barrier and its convenience as an oral treatment, often combined with other agents to enhance efficacy.
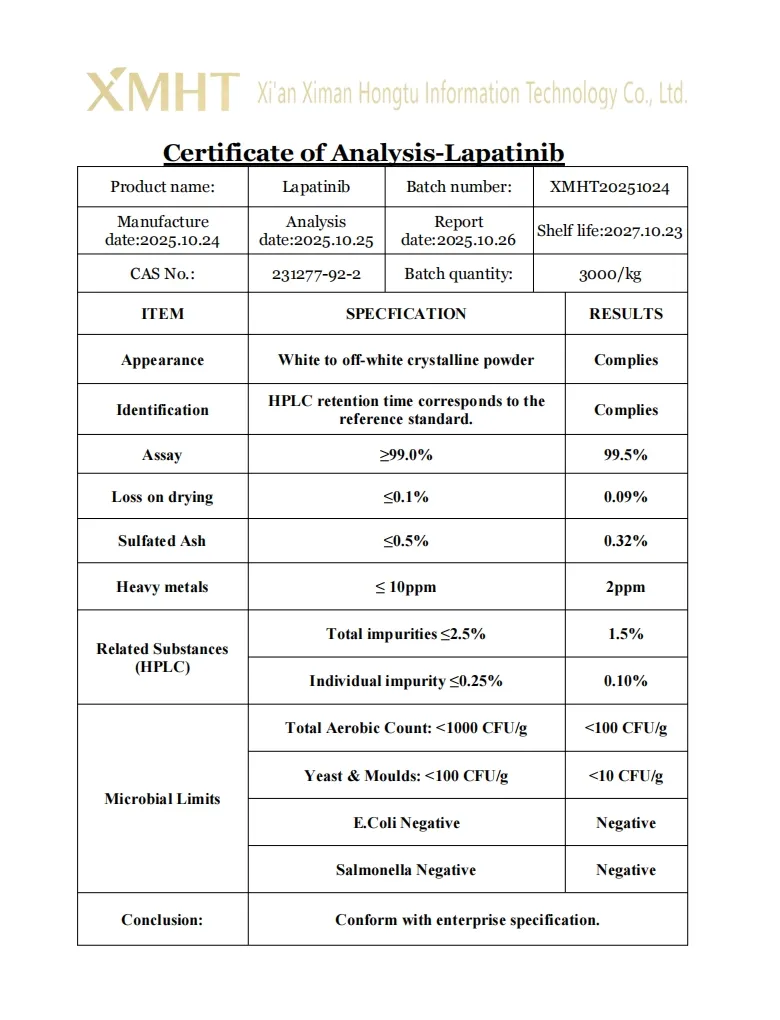





A: Our Lapatinib powder has a purity of over 99%, verified by HPLC testing to ensure pharmaceutical grade quality.
A: It should be stored in a cool, dry place away from direct light. The shelf life is 2 years when stored properly.
A: Yes, research indicates that Lapatinib has the ability to cross the blood-brain barrier, making it relevant for studies involving brain metastases.
A: We follow GMP, ISO 9001 and USP/BP standards. Each batch is provided with a Certificate of Analysis (COA).
A: Our current production capacity for Lapatinib is approximately 3000kg per month.
A: Yes, transport packaging is negotiable to meet specific customer requirements and ensure safe delivery.
 SJ medical
SJ medical