




1 / 5










| Still deciding? Get samples of $ ! US$ 200/kg Request Sample |

This product is typically formulated in a fixed dose combination with another DAA, Sofosbuvir. The synthesis process of Vepatasvir is complex, with extremely strict requirements for purity, crystal form, and related substance control, making it a key raw material for ensuring the safety and effectiveness of the final drug.



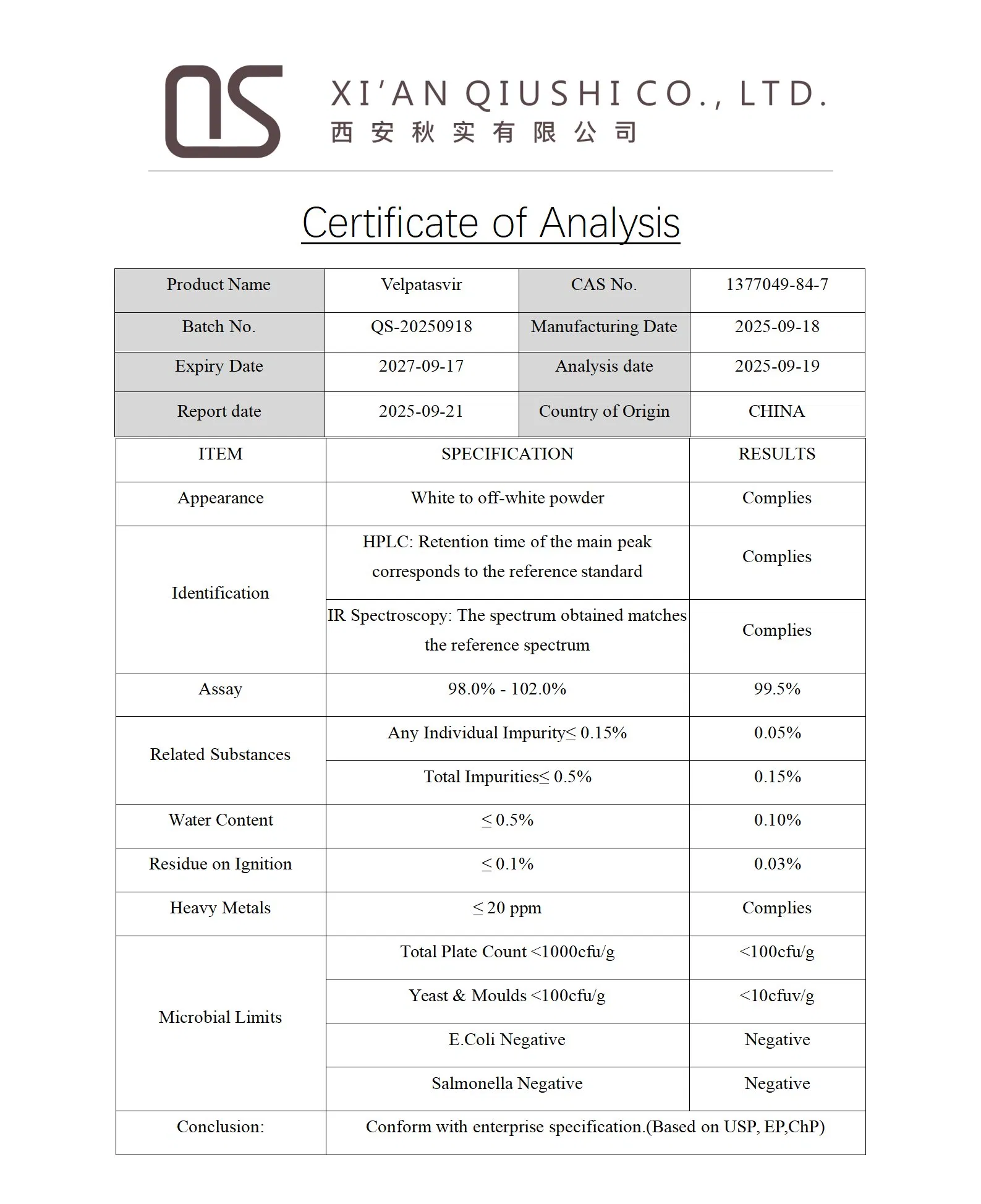
| Product Name | Velpatasvir |
| Appearance | White to off-white powder |
| CAS No. | 1377049-84-7 |
| MF | C49H54N8O8 |
| MW | 883.002 |
The main function of Vepatasvir is to inhibit the function of HCV NS5A protein, which is crucial for the replication of viral RNA and the assembly of viral particles. By tightly binding to NS5A, Vepatasvir can effectively disrupt the formation of HCV replication complexes, thereby strongly inhibiting virus replication.









 SJ medical
SJ medical