




1 / 5










| Still deciding? Get samples of $ ! US$ 35/g Request Sample |

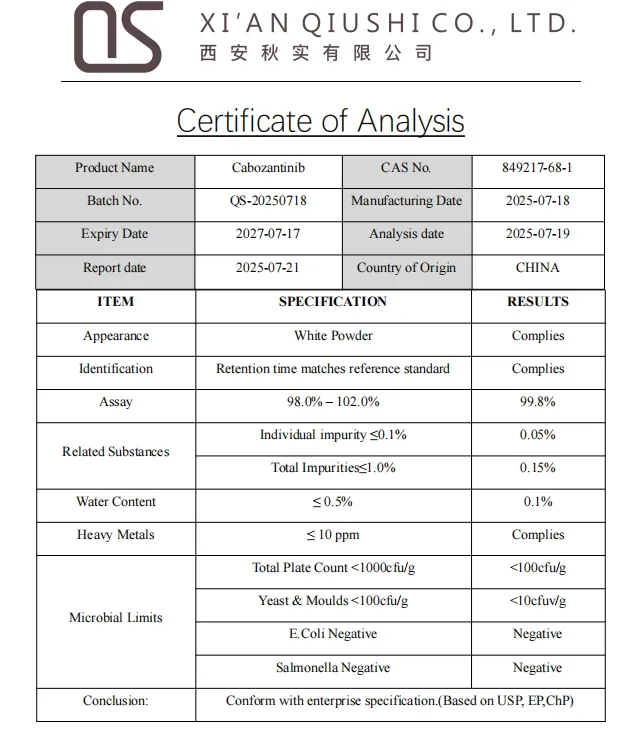
| Parameter | Specification |
| Product Name | Cabozantinib |
| Appearance | White Powder |
| CAS No. | 849217-68-1 |
| Molecular Formula | C28H24FN3O5 |
| Molecular Weight | 501.514 |








 SJ medical
SJ medical