📋
Basic Information
| Product Name |
Afatinib |
| CAS Number |
850140-72-6 |
| Molecular Formula |
C24H25ClFN5O3 |
| Molecular Weight |
485.9 |
| Purity |
>99% |
| Appearance |
White to off-White Crystalline Powder |
| Certification |
GMP, HSE, ISO 9001, USP, BP |
| Test Method |
HPLC |
🔍
Product Description
Afatinib is a potent substance used to treat specific types of malignant tumors, belonging to the class of epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors. At room temperature, it typically appears as a white to pale yellow powder, which is odorless and easily soluble in dimethyl sulfoxide. This substance irreversibly inhibits the activity of EGFR and its related family members, effectively blocking signaling pathways for tumor cell growth and proliferation.
💊
Application & Function
Afatinib is an oral irreversible epidermal receptor (EGFR) tyrosine kinase inhibitor (TKI), primarily used for the treatment of non-small cell lung cancer (NSCLC) harboring specific EGFR gene mutations. As a second-generation EGFR-TKI, it irreversibly binds to the tyrosine kinase domains of the EGFR family (including EGFR/HER1, HER2, HER4).
In clinical practice, it is utilized as a first-line treatment for locally advanced or metastatic NSCLC with EGFR-sensitive mutations (such as exon 19 deletions or exon 21 L858R mutations). It also shows therapeutic effects for squamous cell NSCLC that has progressed after platinum-containing chemotherapy, significantly prolonging progression-free survival and improving patients' quality of life.
📜
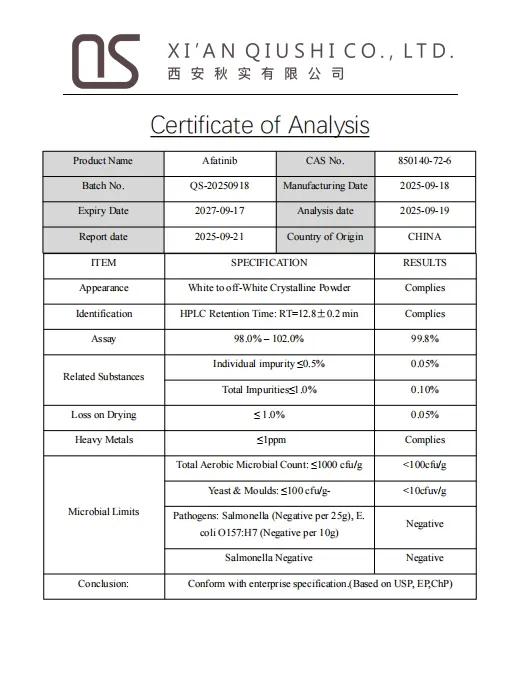
Specifications & Quality Control
📸
Product Display
Frequently Asked Questions
Q1: What is the primary application of Afatinib?
Afatinib is primarily used as a targeted therapy for non-small cell lung cancer (NSCLC), specifically for patients with EGFR gene mutations and those who have progressed after platinum-based chemotherapy.
Q2: What is the purity level of the Afatinib powder?
The product features a high purity level of over 99%, as verified by HPLC testing methods.
Q3: How does Afatinib differ from first-generation TKIs?
Afatinib is a second-generation TKI that binds irreversibly to EGFR family receptors (HER1, HER2, HER4), providing a more sustained inhibition of signaling pathways compared to reversible first-generation inhibitors.
Q4: What are the physical properties of the substance?
It is a white to off-white crystalline powder with a molecular weight of 485.9 and a melting point around 102°C.
Q5: Is a Certificate of Analysis (COA) available?
Yes, every batch is strictly tested, and a detailed Certificate of Analysis (COA) is provided to ensure quality and compliance with standards like USP and BP.
Q6: What certifications does the manufacturing process hold?
The production process complies with international standards, including GMP, ISO 9001, and HSE certifications.

























 SJ medical
SJ medical