














| Still deciding? Get samples of $ ! US$ 190/g Request Sample |

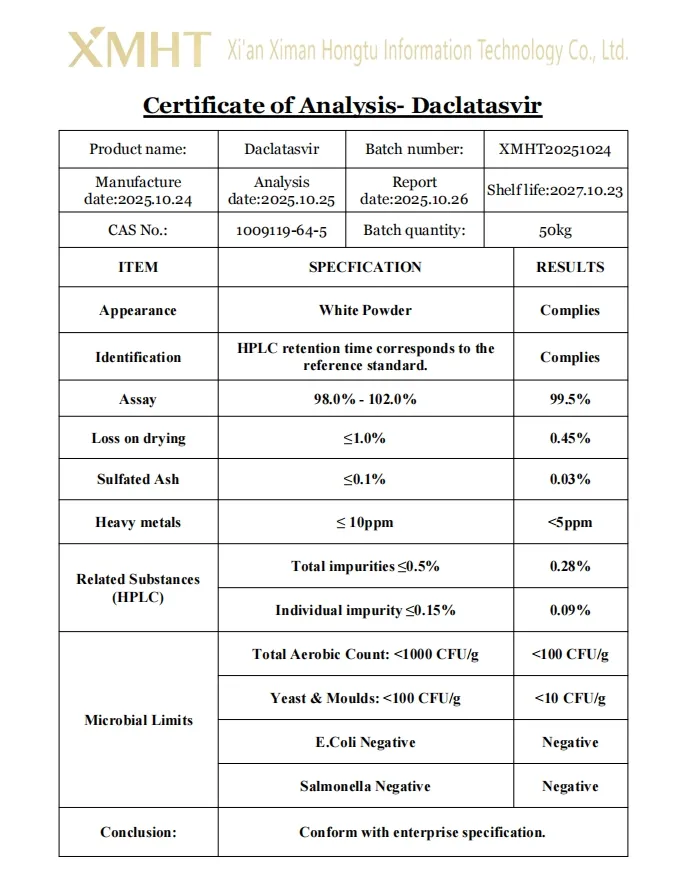
Daclatasvir dihydrochloride is a potent hepatitis C virus NS5A inhibitor. It appears as a white to off-white crystalline powder with a molecular weight of 813.8 g/mol (as dihydrochloride). The compound is hygroscopic and requires protection from moisture. Identification is confirmed by High Performance Liquid Chromatography (HPLC) and Infrared (IR) spectroscopy for structural verification. The purity assessment includes rigorous testing for related substances, process-related impurities, and degradation products. Residual solvents are controlled according to ICH Q3C guidelines, and elemental impurities meet ICH Q3D requirements.


| Parameter | Details |
|---|---|
| Product Name | Daclatasvir |
| Appearance | White Powder |
| CAS Number | 1009119-64-5 |
| Molecular Formula | C40H50N8O6 |
| Molecular Weight | 738.875 |
Daclatasvir functions as a potent and selective inhibitor of the hepatitis C virus NS5A protein. It binds to the N-terminal domain of NS5A, disrupting its multiple functions in viral replication and assembly. The drug inhibits both hyperphosphorylation and hypophosphorylation of NS5A, affecting the formation of the viral replication complex. It demonstrates broad-spectrum activity across multiple HCV genotypes, including genotypes 1 through 6. Daclatasvir has favorable pharmacokinetics allowing once-daily dosing and shows minimal drug-drug interactions.







 SJ medical
SJ medical