📋 Basic Information
Model NO.:
XMHT-Dacomitinib
Certification:
GMP, HSE, ISO 9001, USP, BP
Appearance:
White Crystalline Powder
Grade:
Pharmaceutical Grade
💊 Product Description
| Product name | Dacomitinib |
| Appearance | White Powder |
| CAS NO. | 1110813-31-4 |
| MF | C24H25ClFN5O2 |
| MW | 469.945 |
Dacomitinib, sold under the brand name Vizimpro, is a second-generation oral irreversible pan-HER tyrosine kinase inhibitor. Approved by the US FDA, it is indicated as the first-line treatment for adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) whose tumors are confirmed to have EGFR exon 19 deletion or exon 21 L858R substitution mutation. By virtue of its irreversible binding to EGFR, HER2 and HER4 receptors, it continuously blocks tumor growth signaling pathways.
📊 Application & Function
Dacomitinib is an antitumor compound with a targeted mechanism of action, inhibiting tumor cell proliferation and invasion. It blocks relevant signaling pathways, suppressing the growth, division, and angiogenesis of tumor cells while inducing apoptosis. It features a clear mechanism of action, effectively delaying tumor progression and focusing on disrupting pathological processes without significantly interfering with normal cells.
✅ Specifications & Gallery
❓ Frequently Asked Questions
1. What is the purity of your Dacomitinib powder?
Our Dacomitinib powder maintains a high purity of >99% as verified by HPLC testing Method.
2. For which conditions is Dacomitinib typically indicated?
It is primarily used as a first-line treatment for adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with specific EGFR mutations.
3. What certifications do your products hold?
Our products are manufactured under GMP, ISO 9001 standards and comply with USP and BP specifications.
4. How should Dacomitinib be stored?
It should be stored in a cool, dry place. The shelf life of our powder is 2 years under proper storage conditions.
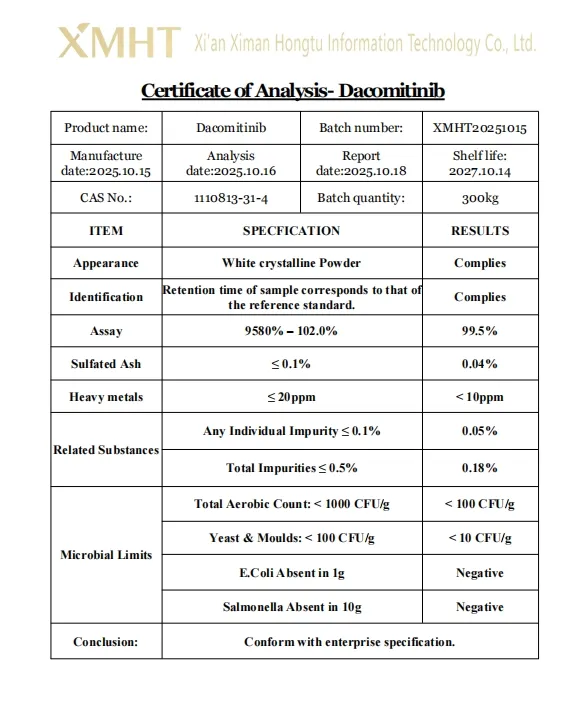
5. Is it possible to provide a Certificate of Analysis (COA)?
Yes, we provide COA and HPLC test reports for every batch to ensure quality and compliance.
6. What is the production capacity for this material?
We have a stable production capacity of approximately 300kg per month to meet both small and large scale requirements.

























 SJ medical
SJ medical