Basic Information
Certification
GMP, HSE, ISO 9001, USP, BP
Grade
Pharmaceutical Grade
Product Description
Dasatinib (CAS number 302962-49-8) is an oral tyrosine kinase inhibitor small molecule targeted drug. It is a white to off white powder and belongs to the class of amino pyrimidine derivatives in terms of chemical structure. This product is prepared through high-precision synthesis and purification processes, and must comply with strict pharmacopoeial standards (such as USP, ChP) to ensure high purity and quality stability in key indicators such as content, related substances, and residual solvents. It is mainly used for the preparation of anti-tumor preparations.
Product Details
| Product Name | Dasatinib |
| Appearance | White crystalline powder |
| CAS | 302962-49-8 |
| MF | C22H26ClN7O2S |
| MW | 488.007 |
Application & Function
Dasatinib is an orally administered agent that exerts its therapeutic effects by targeting specific signaling pathways involved in the proliferation and survival of abnormal cells. It binds to relevant molecular targets to inhibit their activity and block the growth signals that drive the uncontrolled division of affected cells. It is widely used in the clinical management of specific conditions, helping to control disease-related symptoms, improve the clinical status of patients, and enhance their quality of life. Its oral formulation offers convenience for long-term administration.
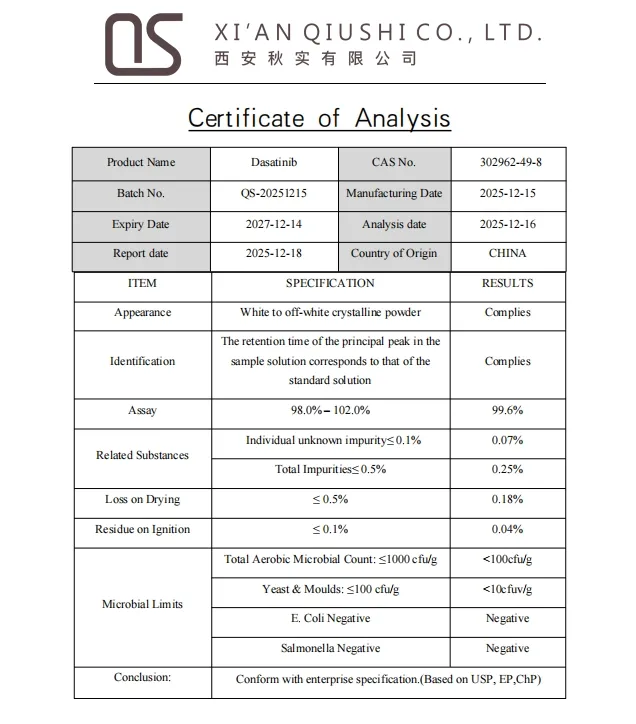
Certificate of Analysis
Product Showcase
Frequently Asked Questions
Q1: What is the purity level of your Dasatinib powder?
Our Dasatinib powder is pharmaceutical grade with a purity level exceeding 99%, tested via HPLC.
Q2: What are the storage requirements for this product?
It should be stored in a cool, dry place away from direct sunlight. The shelf life is 2 years under proper conditions.
Q3: Does the product come with a Certificate of Analysis (COA)?
Yes, every batch is strictly tested and accompanied by a detailed COA to ensure quality standards.
Q4: What is the appearance of Dasatinib?
It typically appears as a white to off-white crystalline powder.
Q5: What certifications does your facility hold?
Our production processes are compliant with GMP, ISO 9001, and HSE standards to ensure global pharmaceutical requirements.
Q6: What is the lead time for large orders?
With a production capacity of 500kg per month, we can accommodate various order sizes with negotiable transport timelines.

























 SJ medical
SJ medical