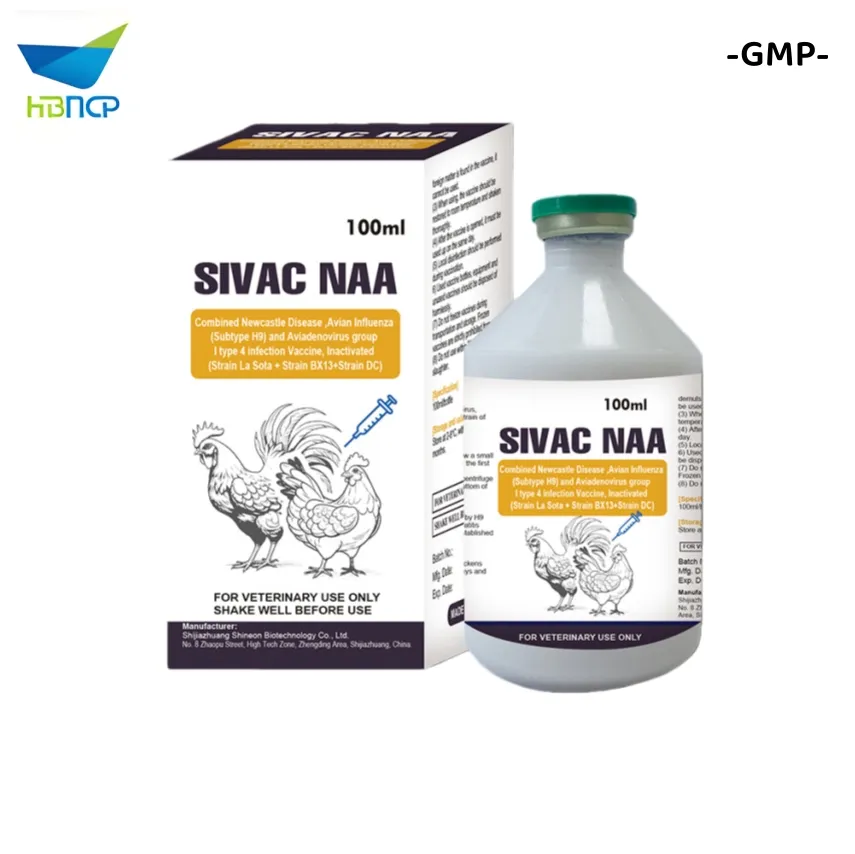
GMP Manufacturer OEM Veterinary Grade Newcastle Disease, Infectious Bronchitis, Avian Influenza H9 subtype and Infectious Bursal Disease Vaccine
📊
Basic Information
Model NO.CH-VACCINE10
TypeThe First Class
FactorsAnimal Species
StoragePrevent High/Low Temp
Veterinary Reg. No.030296176
Transport Package50ml - 500ml
Specification5mg/mL 10mg/mL
TrademarkHBNCP
OriginChina
Production Capacity20000 Bottles/Day
🧪
Main Components & Content
Each milliliter of the vaccine contains the following inactivated antigens and protein:
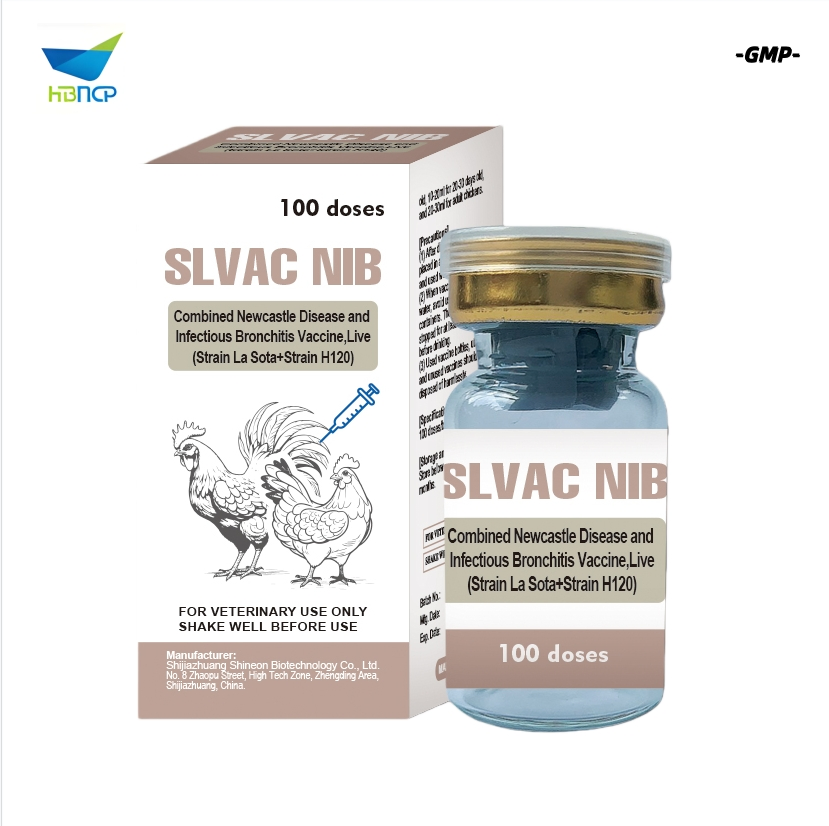
- Newcastle Disease Virus (NDV) Strain La Sota: Viral titer ≥ 108.5 EID50/0.1ml. High safety for all chicken breeds.
- Infectious Bronchitis Virus (IBV) Strain M41: Viral titer ≥ 107.2 EID50/0.2ml. Prevents respiratory lesions.
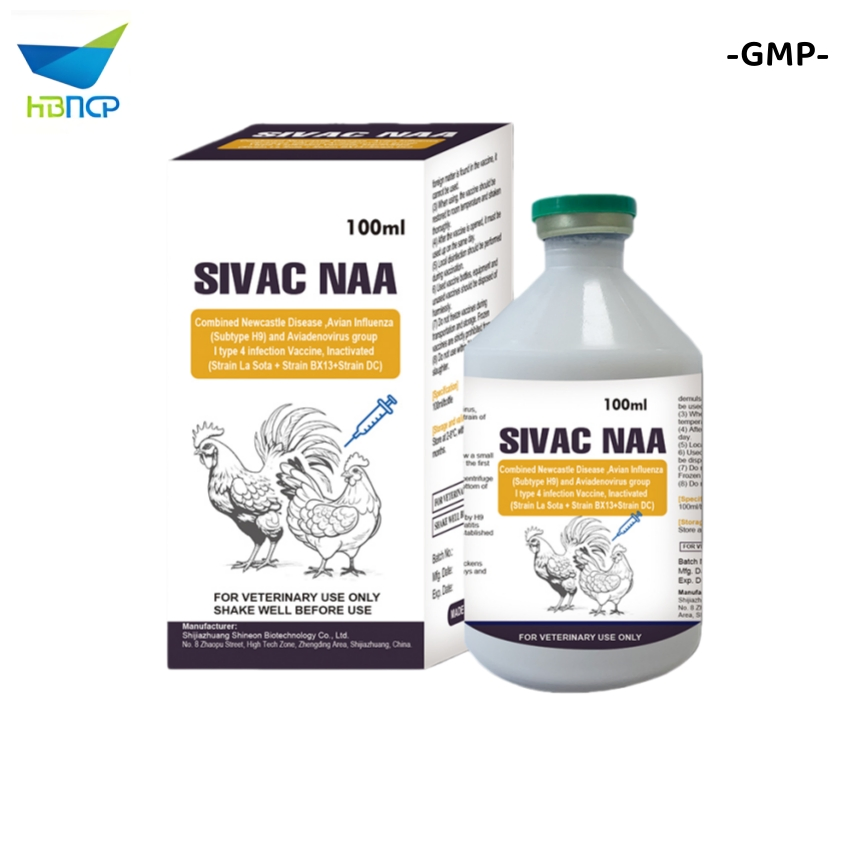
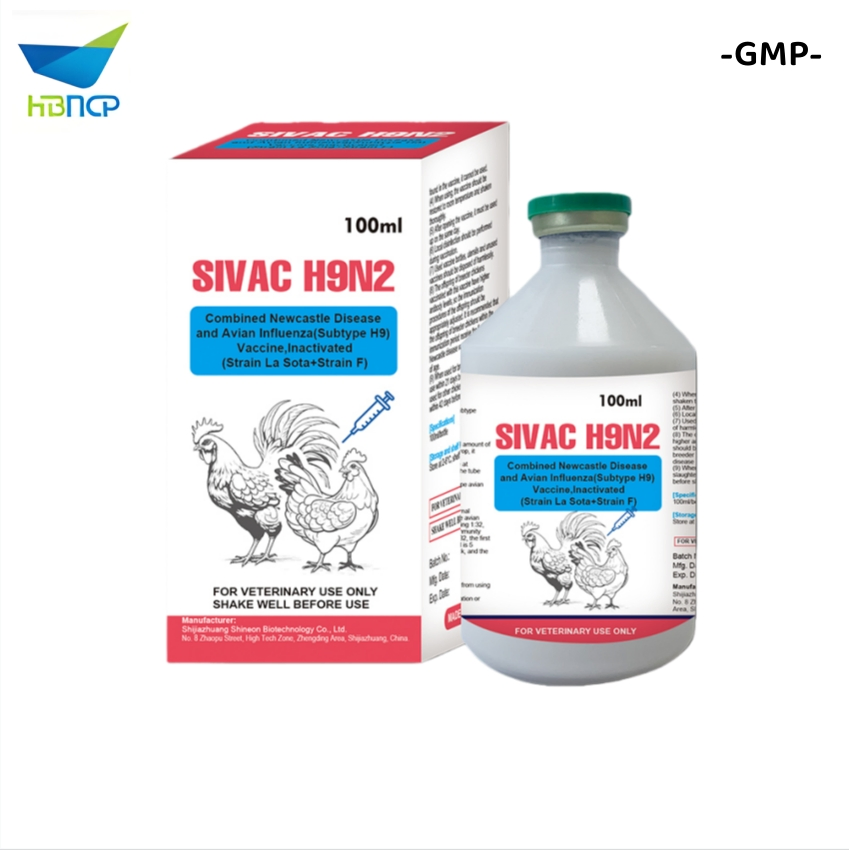
- Avian Influenza Virus (AIV) H9 Subtype Strain SZ: Viral titer ≥ 108.5 EID50/0.1ml. Broad immune protection spectrum.
- Infectious Bursal Disease Virus (IBDV) Recombinant VP2: AGD titer ≥ 1:5.12. High purity and stable immunogenicity.
Excipients include white oil, emulsifier, and stabilizer, complying with veterinary drug standards.
💉
Indications & Administration
Indications: Used for the prevention of Newcastle Disease, Infectious Bronchitis, H9 subtype Avian Influenza and Infectious Bursal Disease. It realizes "one injection for four diseases".
Dosage:
• 1-4 week-old chickens: 0.3ml per chicken.
• Over 4 weeks old: 0.5ml per chicken.
Route: Subcutaneous injection in the neck or intramuscular injection in the chest.
🏭
Manufacturing & Quality
Equipped with high-performance liquid chromatographs, UV-visible spectrophotometer, gas chromatograph, and bacterial endotoxin analyzer to ensure stability and safety.
❓
Frequently Asked Questions
Does the product have compliance qualifications for export?
Yes, all products have obtained GMP certification and necessary "Veterinary Drug Production Licenses". We provide COA, Free Sale Certificates (FSC), and Certificates of Origin for customs clearance.
What quality standards are followed?
Our products are strictly produced according to the standards of the Chinese Veterinary Pharmacopoeia and undergo rigorous batch testing.
What are the storage and transportation requirements?
The vaccine must be stored and transported at 2-8ºC in a dark place. It is vital to avoid freezing, high temperatures, and severe vibration.
How long is the vaccine valid after opening?
While the total shelf life is 24 months, it should be used within 7 days once the bottle is opened, provided it is kept in a refrigerator at 2-8ºC.
What is the immune protection period?
Protective antibodies are produced 21 days after vaccination. The period is 3 months for chickens under 4 weeks old and 4 months for those over 4 weeks old.
Can the packaging be customized for OEM orders?
Yes, we offer customizable specifications including 100ml, 250ml, 500ml, and 1000ml bottles to meet OEM requirements.
























 SJ medical
SJ medical