




1 / 5










| Still deciding? Get samples of $ ! US$ 115/Piece Request Sample |
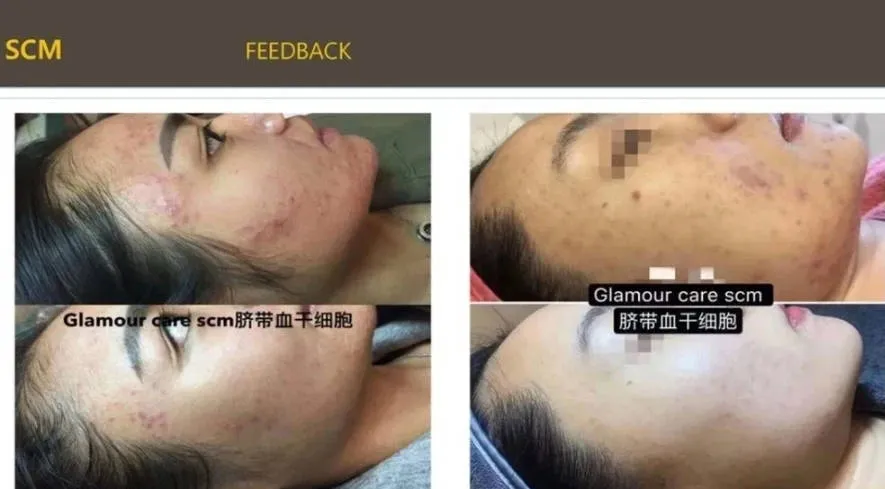
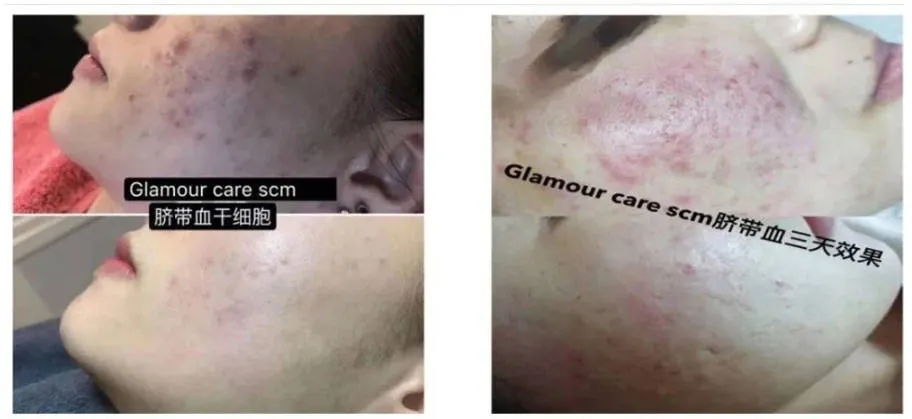
Korean SCM umbilical cord blood stem cell hydrography Acne removal



Korean SCM umbilical cord blood stem cell hydrogel is a product with the ability to self renew and differentiate cells, known as the "universal assistant of the skin" in the medical field.
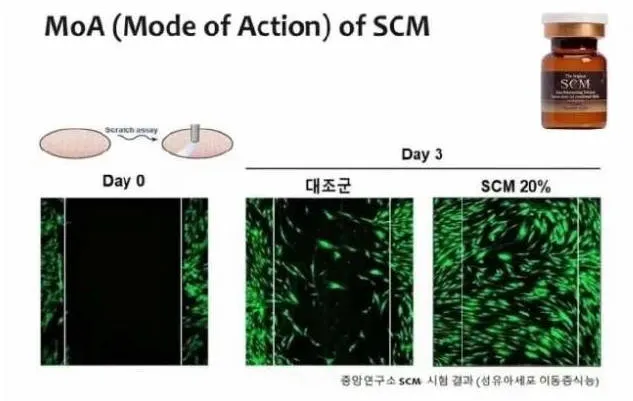
| Extracellular Matrix Proteins (ECM) | Increases binding force between skin cells, helping maintain skin elasticity and moisturizing by holding water inside the skin. |
| ADSCs | SCM adipose stem cells secrete rich growth factors (VEGF, FGF, HGF, TGF-β) and ECM proteins like collagen and hyaluronic acid to promote new cell growth. |
| Antioxidant Enzymes | Contains various cytokines and antioxidant enzymes that prevent cell membrane hardening and inhibit skin aging. |
Treatment Notice: Patients must be informed about indications and potential reactions. Must be used on healthy, disinfected skin by licensed professionals.

 SJ medical
SJ medical