












| Still deciding? Get samples of $ ! US$ 0/Piece Request Sample |
This Malaria P.f/P.v Rapid Test Kit is a rapid test for the qualitative detection of Malaria Antigen P.f or/and P.v in human venous and capillary whole blood. For professional use only.
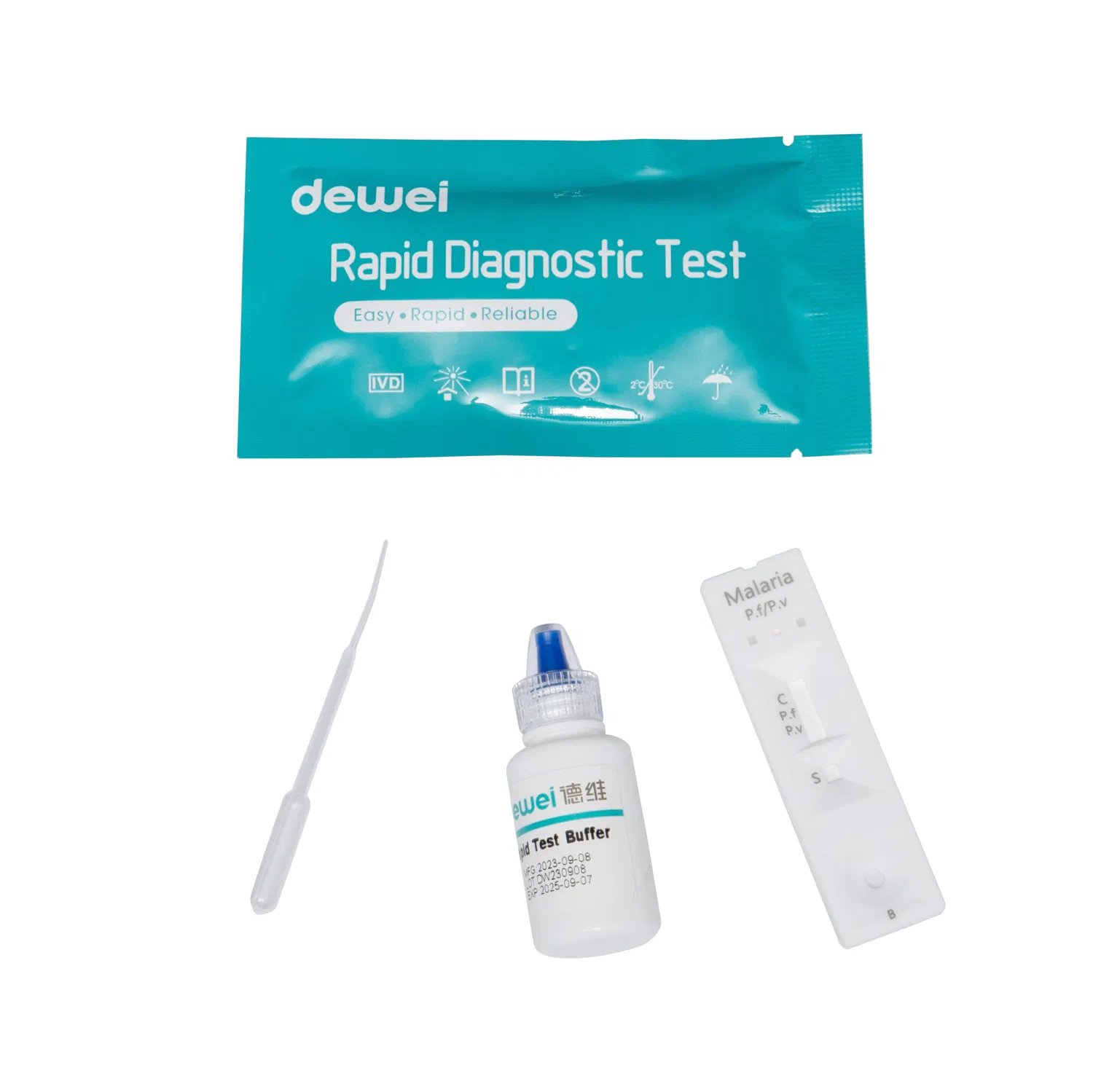
| Component | Rapid Test + Buffer + Pipettes |
| Principle | Colloidal gold rapid tests |
| Reading Time | Within 15mins |
This test targets the histidine-rich protein II (HRPII) antigen specific to Plasmodium falciparum (P.f.) and a p.v-malarial antigen common to P. vivax (P.v.).
1. Preparation: Allow the device and specimen to reach room temperature (15-30°C).
2. Specimen Transfer:
• Pipette: Transfer 5 mL whole blood to Well-S, then add 3~4 full drops of buffer to Well-B.
• Disposable Pipette: Draw blood to Fill Line, transfer to Well-S, then add 3~4 drops of buffer to Well-B.
3. Result: Wait for colored lines. Read at 15 minutes. Do not interpret after 20 minutes.
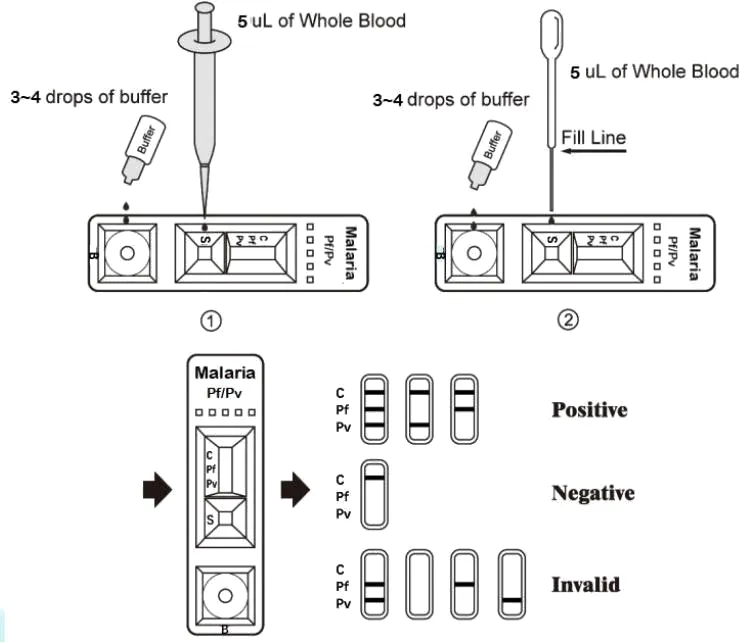
✅ Positive (+): Lines appear in both Control (C) and specific Test regions (Pf/Pv).
❌ Negative (-): Only the Control line (C) appears.
⚠️ Invalid: No Control line (C) appears. Discard and repeat with new test.


R&D TEAM

PRODUCTION LINE

CERTIFICATES

EXHIBITIONS & SHIPMENTS


 SJ medical
SJ medical