




1 / 5










| Still deciding? Get samples of $ ! US$ 3700/kg Request Sample |

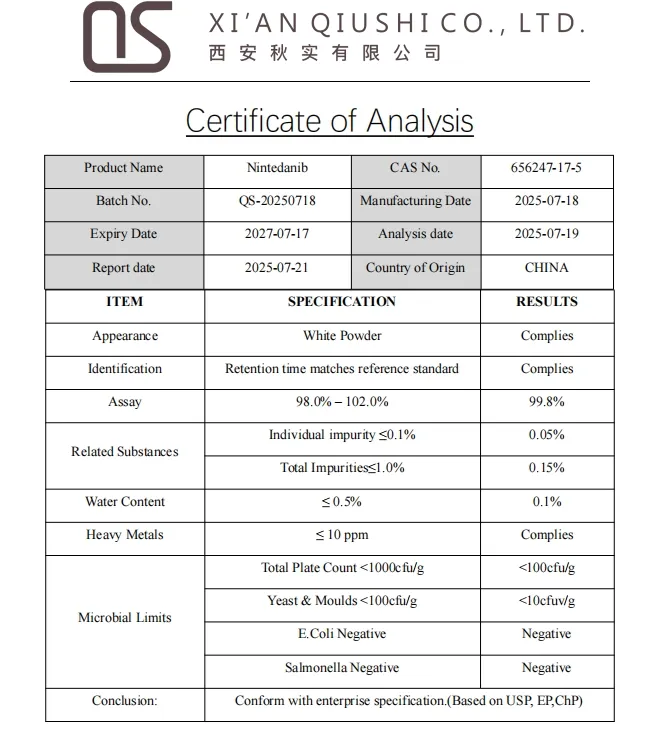
| Model NO. | QS-Nintedanib |
|---|---|
| Certification | GMP, HSE, ISO 9001, USP, BP |
| Suitable for | Elderly, Children, Adult |
| State | Powder |
| Purity | >99% |
| CAS No. | 656247-17-5 |
| Molecular Formula | C31H33N5O4 |
| Molecular Weight | 539.62482 |
| Appearance | White Powder |
| Assay | 99% |
| Test Method | HPLC |













 SJ medical
SJ medical