
Anti-Tumor Material Oxaliplatin Powder
Basic Information
Model NO.XMHT Oxaliplatin
CertificationGMP, HSE, ISO 9001, USP, BP
StatePowder
Purity>99%
CAS No.61825-94-3
MFC8H12N2O4Pt
AppearanceWhite or White Crystal Powder
GradePharmaceutical Grade
Assay99.6%
Storage Temp2-8°C
Product Description
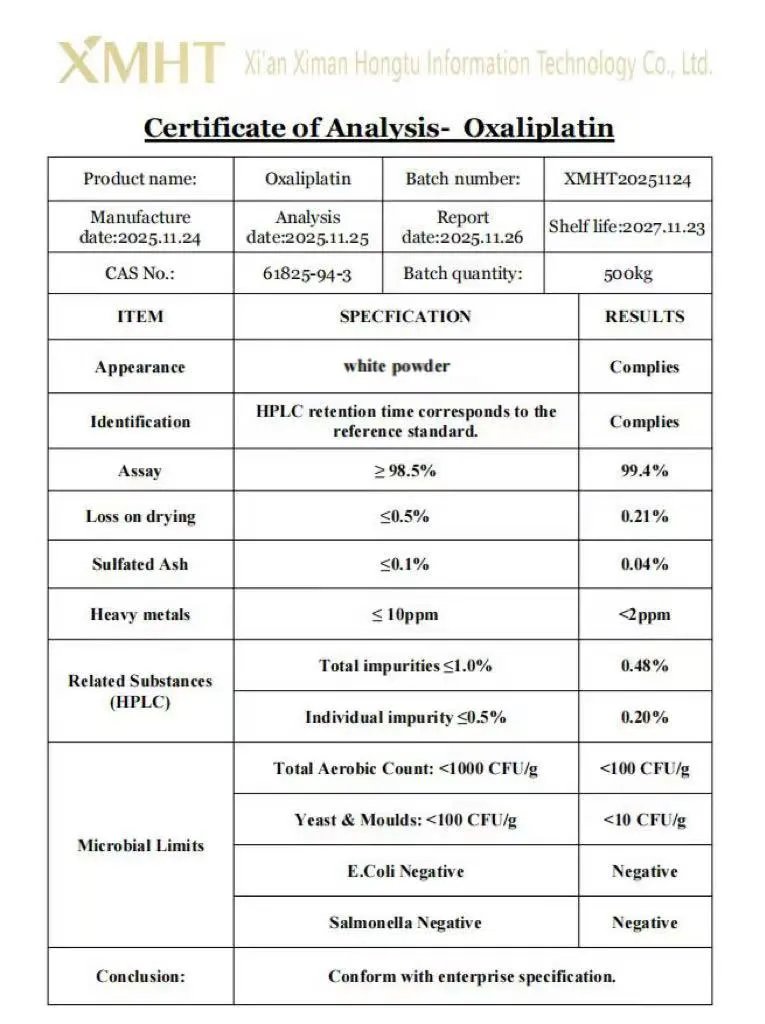
Oxaliplatin is a platinum-based antineoplastic agent with the chemical name cis-oxalato(trans-l-1,2-diaminocyclohexane)platinum(II). It appears as a white to off-white crystalline powder and is moderately soluble in water. Identification is typically performed by infrared spectroscopy (IR) and high-performance liquid chromatography (HPLC). Purity is assessed with a specification of not less than 98.0 percent. The product is light-sensitive and requires storage in the dark at controlled room temperature or under refrigeration.
Product Details
| Product Name | Oxaliplatin |
| Appearance | White Powder |
| CAS | 61825-94-3 |
| MF | C8H12N2O4Pt |
| MW | 395.28 |

Application & Function
Oxaliplatin functions as a third-generation platinum-based chemotherapy drug. Primary mechanism involves the formation of covalent DNA adducts, leading to inhibition of DNA replication and transcription, ultimately triggering apoptosis in cancer cells. It is commonly used in combination regimens such as FOLFOX and CAPOX for treating colorectal, gastric, and pancreatic cancers. Unlike cisplatin, its unique structure allows it to bypass certain DNA repair mechanisms.
Technical Specifications
Frequently Asked Questions
Q1: What is the purity level of your Oxaliplatin powder?
Our Oxaliplatin powder maintains a purity level of over 99%, as tested by HPLC methods.
Q2: How should the product be stored to maintain stability?
It should be stored at 2-8°C, protected from light and moisture, to ensure a shelf life of up to 2 years.
Q3: Which certifications does this product hold?
The product is manufactured under GMP, ISO, and HSE certifications, meeting USP and BP standards.
Q4: What are the primary clinical applications?
Oxaliplatin is primarily used in chemotherapy for colorectal cancer, gastric cancer, and other gastrointestinal malignancies.
Q5: Is this product suitable for pharmaceutical formulation?
Yes, it is pharmaceutical grade material suitable for use as an active pharmaceutical ingredient (API).
Q6: What is the appearance of the material?
It is a white or white crystalline powder, providing excellent solubility for medical manufacturing.
























 SJ medical
SJ medical