🔷 Pharmaceutical Material Temozolomide Powder Antineoplastic
📋 Basic Information
Model NO.
XMHT- Temozolomide
Certification
GMP, HSE, ISO 9001, USP, BP
Molecular Formula
C6H6N6O2
Grade
Pharmaceutical Grade
🔍 Product Description
Temozolomide is an oral alkylating antineoplastic agent that has become a first-line standard of care for high-grade gliomas, such as glioblastoma multiforme, due to its proven efficacy and excellent bioavailability. Functioning as a prodrug, it undergoes spontaneous conversion in the body to the active metabolite MTIC, which can cross the blood-brain barrier. Its primary mechanism involves methylating DNA at specific sites, which introduces damage that disrupts DNA replication and ultimately triggers apoptosis in tumor cells.
🔬 Product Details
| Product Name | Temozolomide |
| Appearance | White Powder |
| CAS | 85622-93-1 |
| Molecular Weight | 194.15084 |
🚀 Application & Function
One of temozolomide's distinguishing characteristics is its high oral bioavailability and capacity to cross the blood-brain barrier, achieving therapeutic concentrations in the central nervous system. Beyond high-grade gliomas, temozolomide demonstrates activity in other solid tumors and hematologic malignancies, including metastatic melanoma and neuroendocrine tumors. Research continues to explore temozolomide's synergy with other therapeutic modalities, including immunotherapy and novel DNA repair inhibitors.
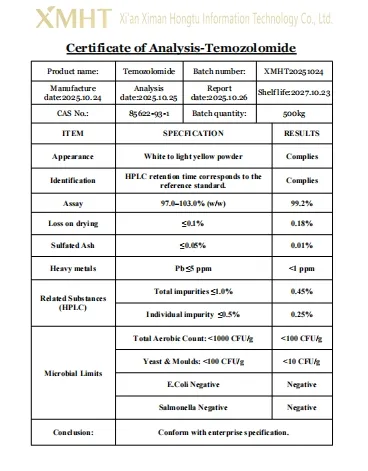
📊 Specifications & Certificates
❓ Frequently Asked Questions
What is the purity level of Temozolomide powder?
Our Temozolomide powder is pharmaceutical grade with a purity level exceeding 99% as verified by HPLC.
What are the primary applications of this product?
It is primarily used as an antineoplastic agent in pharmaceutical research and production, specifically for treating high-grade gliomas and glioblastoma multiforme.
Can Temozolomide cross the blood-brain barrier?
Yes, one of its key features is its high oral bioavailability and its ability to effectively cross the blood-brain barrier.
What are the storage requirements and shelf life?
The product has a shelf life of 2 years when stored under proper pharmaceutical conditions.
Does the product meet international standards?
Yes, our product complies with GMP, HSE, ISO 9001, USP, and BP standards.
What is the production capacity for this material?
We have a stable production capacity of approximately 500kg per month to meet industrial demands.























 SJ medical
SJ medical