📋 Basic Information
Model NO.
XMHT-Temozolomide
Certification
GMP, HSE, ISO 9001, USP, BP
Grade
Pharmaceutical Grade
🔍 Product Description
Temozolomide is an oral alkylating antineoplastic agent that has become a first-line standard of care for high-grade gliomas, such as glioblastoma multiforme, due to its proven efficacy and excellent bioavailability. Functioning as a prodrug, it undergoes spontaneous conversion in the body to the active metabolite MTIC, which can cross the blood-brain barrier.
🔬 Product Details
| Product Name | Temozolomide |
| Appearance | White Powder |
| CAS | 85622-93-1 |
| Molecular Formula | C6H6N6O2 |
| Molecular Weight | 194.15084 |
🚀 Application & Function
One of temozolomide's distinguishing characteristics is its high oral bioavailability and capacity to cross the blood-brain barrier, achieving therapeutic concentrations in the central nervous system. Beyond high-grade gliomas, temozolomide demonstrates activity in other solid tumors and hematologic malignancies, including metastatic melanoma and neuroendocrine tumors.
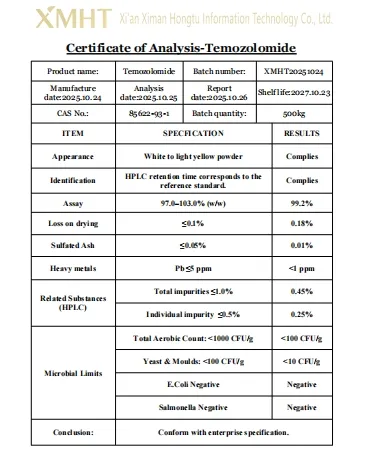
📊 Technical Specifications
Frequently Asked Questions
🔹 What is the purity level of Temozolomide powder?
The purity of our Temozolomide powder is ≥99% as verified by HPLC testing.
🔹 Can Temozolomide cross the blood-brain barrier?
Yes, its high lipophilicity and unique chemical structure allow it to cross the blood-brain barrier effectively.
🔹 What is the typical shelf life?
The shelf life is 2 years when stored under appropriate cool and dry conditions.
🔹 What certifications does this product hold?
It is produced under GMP, ISO 9001 standards and complies with USP/BP pharmaceutical grades.
🔹 What is the appearance of the product?
It appears as a high-quality white crystalline powder.
🔹 How is the product transported?
Packaging and transport are negotiable to ensure safety and stability during transit.






















 SJ medical
SJ medical