Basic Information
Model NO.:QS-Trametinib
Certification:GMP, HSE, ISO 9001, USP, BP
Suitable for:Elderly, Children, Adult
State:Powder
Purity:>99%
CAS:871700-17-3
MF:C26H23FIN5O4
Appearance:White Powder
Capacity:1000kg/Month
Product Description
Trametinib is an oral MEK (mitogen-activated protein kinase kinase) inhibitor-targeted antineoplastic drug. Its core mechanism of action is to specifically inhibit the activity of MEK1/2 proteins, blocking the abnormal activation of the RAS-RAF-MEK-ERK signaling pathway. This pathway is often mutated and activated in various tumor cells, leading to uncontrolled proliferation and survival of tumor cells.
Product Details
| Product Name | Trametinib |
| Appearance | White Powder |
| CAS No. | 871700-17-3 |
| Molecular Formula | C26H23FIN5O4 |
| Molecular Weight | 615.402623 |
Application & Function
Trametinib is a selective, oral mitogen-activated protein kinase (MEK) inhibitor, a type of targeted antineoplastic agent primarily used for the treatment of advanced or metastatic cancers harboring activating mutations in the BRAF gene. It specifically binds to and inhibits MEK1/2, blocking the abnormal activation of ERK and interrupting the signaling cascade that promotes tumor cell proliferation.
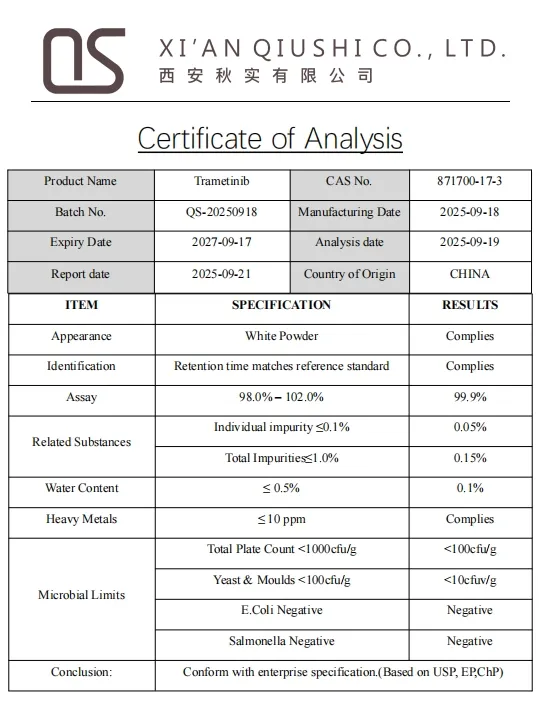
Specification & COA
Frequently Asked Questions
Q1: What is the purity of your Trametinib powder?
A: Our Trametinib API maintains a high purity level of over 99%, as confirmed by HPLC testing.
Q2: What certifications does the product have?
A: The product is manufactured under GMP, HSE, and ISO 9001 standards, complying with USP and BP specifications.
Q3: What is the primary application of Trametinib?
A: It is primarily used as a targeted pharmaceutical agent for advanced or metastatic cancers involving BRAF mutations.
Q4: How is the product transported?
A: We offer flexible and negotiable transport packages to ensure the stability and safety of the powder during transit.
Q5: What is your monthly production capacity?
A: Our current production capacity for Trametinib is approximately 1000kg per month.
Q6: Can I request a Certificate of Analysis (COA)?
A: Yes, every batch is accompanied by a detailed COA and test results from HPLC analysis.

























 SJ medical
SJ medical