



1 / 5










| Customization: | Available |
|---|---|
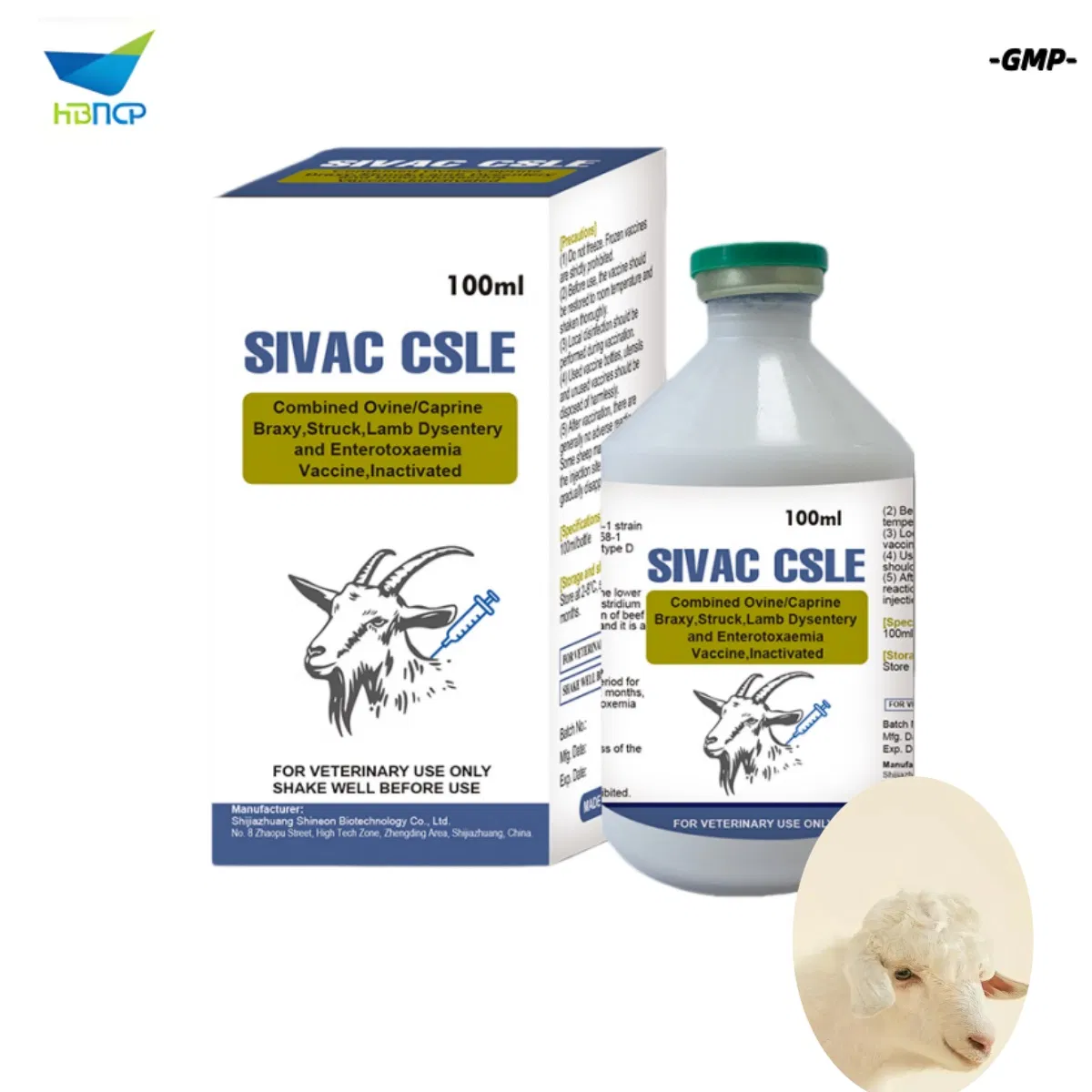
| Varieties: | Infectious Disease Prevention Medicine |
| Component: | Virus |
This product is a combined inactivated vaccine containing antigens from the following Clostridium species, inactivated and adjuvanted.
| Active Ingredient | Target Disease |
|---|---|
| Clostridium septicum (Inactivated Toxoid) | Braxy |
| Clostridium novyi type B (Inactivated Toxoid) | Struck (Black Disease) |
| Clostridium perfringens type B (Inactivated Toxoid) | Lamb Dysentery |
| Clostridium perfringens type D (Inactivated Toxoid) | Enterotoxaemia (Pulpy Kidney) |
| Animal Category | Primary Dose | Booster Dose | Revaccination |
|---|---|---|---|
| Lambs / Kids (< 3m) | 1.0 mL | 1.0 mL (after 3-4 weeks) | Annual 2.0 mL |
| Adult Sheep / Goats | 2.0 mL | Not required if prev. vax | Annual 2.0 mL |
| Pregnant Ewes / Does | 2.0 mL | 4-6 weeks before lambing | Annual 2.0 mL |



Established in 1998, our facility covers over 26,000 square meters with 11 GMP certified workshops and 14 production lines. We utilize high-performance liquid chromatographs, UV-visible spectrophotometers, and gas chromatographs to ensure the highest quality standards.



 SJ medical
SJ medical