









| Customization: | Available |
|---|---|
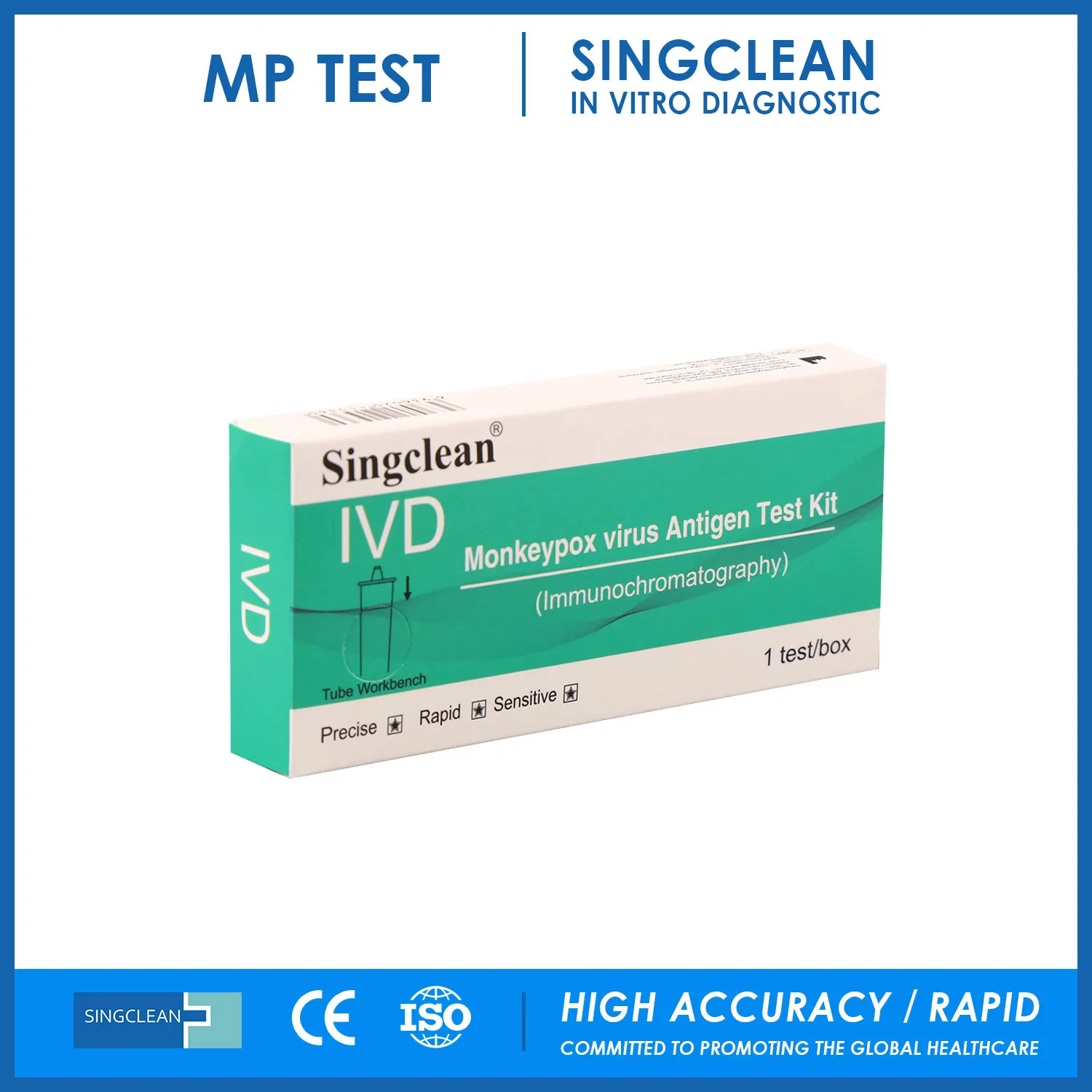
| Type: | IVD Reagent |
| Function: | Malaria Test |

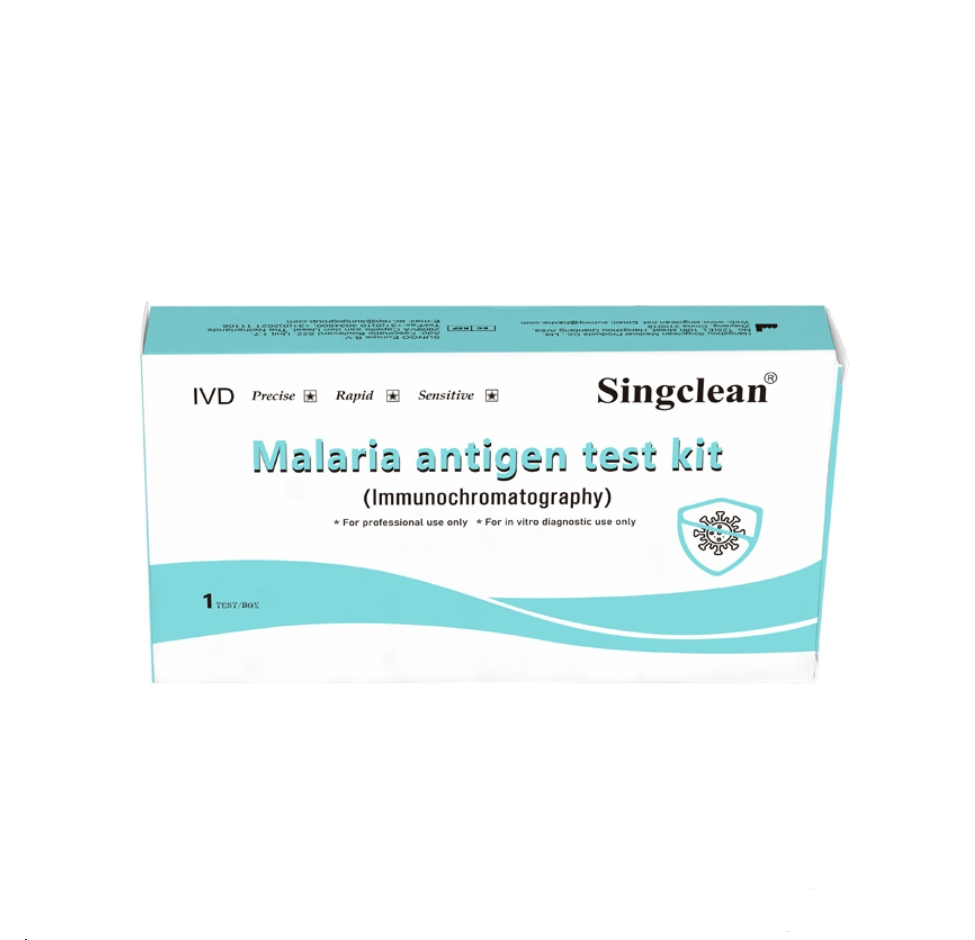
Malaria antigen test kit (Immunochromatography)
Instructions for Use: For professional/in vitro diagnostic use only.
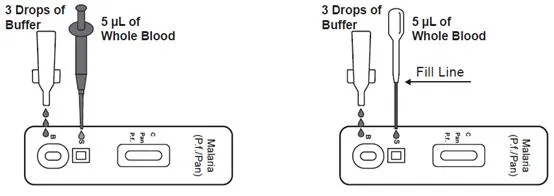
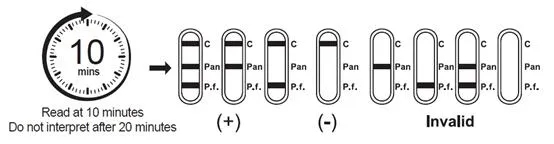
A rapid chromatographic immunoassay for the qualitative detection of circulating plasmodium falciparum (P.f.) and Pan-malaria antigens (P.f., P.v., P.o. and P.m.) in whole blood.
Malaria is one of the world's most prevalent diseases, invading human red blood cells. According to the WHO, the worldwide prevalence is estimated at 300-500 million cases annually. The Malaria antigen test kit is a rapid test to qualitatively detect the presence of P falciparum-specific-HRP and Pan-LDH (Pan) utilizing colloid gold conjugate.
The device uses a membrane pre-coated with anti-HRP-II antibodies and monoclonal antibodies to detect Plasmodium species. A colored line appears in specific regions if antigens are present, while a control line always appears to ensure proper procedure.





 SJ medical
SJ medical